News
Article
Sponsored Article
Understanding Physical and Cognitive Functioning in Patients With Relapsing Multiple Sclerosis
For the estimated 2.3 million people worldwide living with relapsing forms of multiple sclerosis (RMS), cognitive impairment and physical disability can play a prevalent role.1-7
The impact of RMS on physical function is well established. Up to 80% of patients with RMS will experience some form of physical disability within 10 to 15 years of their diagnosis.2 In addition, while further studies are needed, measuring brain volume loss (BVL) may be important to monitoring the condition. Through the use of magnetic resonance imaging (MRI), changes in white and grey matter of the brain can be detected. Researchers previously assumed RMS primarily affected the white matter, but recent research suggests loss of grey matter is the main driver of the pathological disease progression of RMS, making it a clinically relevant target to address overall RMS disease burden.8,9
“The neurodegenerative aspects of RMS caused by brain volume loss have severe clinical implications on cognitive function and day-to-day activities for patients,” said Ralph H.B. Benedict, PhD, a board-certified clinical neuropsychologist and professor of neurology at the State University of New York at Buffalo. “By expanding our understanding of brain volume loss, an opportunity exists to enhance monitoring patients’ conditions and manage them accordingly.”
Physical and cognitive symptoms of RMS
Physical symptoms associated with RMS include difficulty walking, muscle weakness, balance problems and spasticity—which can impact a patient’s ability to work and participate in social events.2,3
Commonly seen cognitive symptoms include slowed processing of information and memory impairment.10,11 About half of patients are affected by cognitive changes, which usually manifest later in the disease.
The impact of physical and cognitive impairment
Mobility impairment is common in patients with RMS. In a 2013 study of 27,918 patients in the North American Research Committee on Multiple Sclerosis registry, 45% of patients with RMS between the ages of 18 and 64 needed a mobility device to help them walk.4 Additionally, 50% of patients said they had difficulty with daily activities 15 years after diagnosis.4
From a cognitive perspective, volume loss of the whole brain and of the thalamus (a grey matter structure) have been correlated with cognitive decline, which can impact patients’ lives.6,12,13 RMS BVL rates are greatest at disease onset, affecting approximately half of patients in the earliest stages of disease, and are greater in younger patients.8,9,14-17
“Cognitive dysfunction is associated with interference with daily tasks, depression and socioeconomic challenges,"10,11 said Dr. Benedict. “Commonly recognized domains of cognitive impairment include diminished cognitive processing speed—the primary factor impacting employment—as well as episodic memory and executive function.”10,18,19
Figure 1
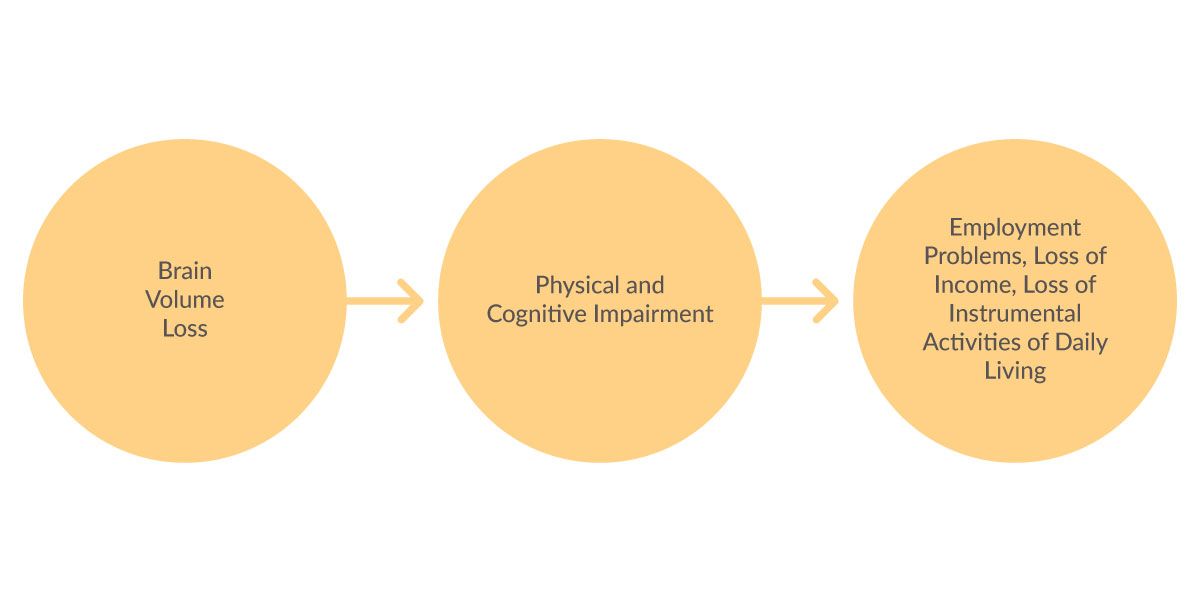
Monitoring RMS
Early detection of cognitive impairment with quantitative measurement tools can help practitioners understand how their patients are progressing.18 In addition to its use in diagnosis and prognosis, MRI is valuable for monitoring purposes in patients with RMS. The presence of active lesions on brain MRI, either at baseline or during the first year of treatment, has been shown to be a useful predictive measure of disease activity.20 In 2018, the National Multiple Sclerosis Society recommended the use of the Symbol Digit Modalities Test to assess cognitive processing speed.10
Physical symptoms and their progression are often measured using the Expanded Disability Status Scale (EDDS). This scale helps quantify the physical effects of RMS and gives healthcare providers a reference from which to monitor changes in function over time.21
One treatment option to consider
One treatment option for patients with RMS is ZEPOSIA (ozanimod), a prescription pill for adults with RMS. ZEPOSIA is indicated for the treatment of RMS, to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.22,23 It is not known if ZEPOSIA is safe and effective in children.23
The efficacy of ZEPOSIA was demonstrated in two randomized, double-blind, double-dummy, parallel-group, active comparator-controlled clinical trials of similar design in patients with RMS. Patients in SUNBEAM (Study 1) were treated until the last enrolled patient completed one year of treatment. Patients in RADIANCE (Study 2) were treated for 24 months. Both studies included patients who had experienced at least one relapse within the prior year or one relapse within the prior two years with evidence of at least a gadolinium-enhancing (GdE) lesion in the prior year and had an EDSS score from 0 to 5.0 at baseline. Patients with primary progressive multiple sclerosis were excluded.23
Patients were randomized to receive either ZEPOSIA 0.92 mg, given orally once daily beginning with a dosage titration or weekly intramuscular AVONEX® (interferon (IFN) beta-1a). Confirmed disability progression was evaluated in a pooled analysis of SUNBEAM and RADIANCE. In SUNBEAM, a total of 895 patients were randomized to receive ZEPOSIA (n=447) or IFN beta-1a (n=448). In RADIANCE, a total of 874 patients were randomized to receive ZEPOSIA (n=433) or IFN beta-1a (n=441).23
SELECTED IMPORTANT SAFETY INFORMATION
ZEPOSIA is associated with the following warnings and precautions: infections, progressive multifocal leukoencephalopathy, bradyarrhythmia and atrioventricular conduction delays, liver injury, fetal risk, increased blood pressure, respiratory effects, macular edema, posterior reversible encephalopathy syndrome, unintended additive immunosuppressive effects from prior treatment with immunosuppressive or immune-modulating drugs, severe increase in multiple sclerosis disability after stopping ZEPOSIA and immune system effects after stopping ZEPOSIA.
Please see below for additional Important Safety Information.
The primary endpoint of both SUNBEAM and RADIANCE was the annualized relapse rate (ARR) over the treatment period (SUNBEAM) and 24 months (RADIANCE). Additional outcome measures included the number of new or enlarging MRI T2 hyperintense lesions over 12 and 24 months, the number of MRI T1 Gadolinium-enhancing (Gd+) lesions at 12 and 24 months, and the time to confirmed disability progression, defined as at least a one-point increase from baseline EDSS, confirmed after 3 months and after 6 months. Results showed powerful efficacy in reducing ARR, GdE brain lesions and new/enlarging T2 lesions vs AVONEX23:
- ZEPOSIA demonstrated a relative reduction in ARR versus AVONEX of 48% through one year and 38% at two years (absolute ARR of 0.18 versus 0.35 and 0.17 versus 0.28, respectively).23
- In a post-hoc analysis, patients not previously treated with a disease-modifying therapy (DMT-naïve) had an ARR of 0.130.24
- In the open-label extension study, patients continuously treated with ZEPOSIA up to six years had an ARR of 0.093.25
- At one year, treatment with ZEPOSIA reduced the number of T1-weighted GdE brain lesions more than AVONEX (0.16 vs 0.43), a relative reduction of 63%, and reduced the number of new or enlarging T2 lesions (1.47 vs 2.84), a relative reduction of 48%.23
- At two years, treatment with ZEPOSIA reduced the number of T1-weighted GdE brain lesions more than AVONEX (0.18 vs 0.37), a relative reduction of 53%. ZEPOSIA also reduced the number of new or enlarging T2 lesions vs AVONEX (1.84 vs 3.18), a relative reduction of 42%.23
Nine of 10 patients showed no confirmed three-month disability progression. There was no statistically significant difference in the three-month and six-month confirmed disability progression between ZEPOSIA and IFN beta-1a-treated patients over two years (7.6% of patients treated with ZEPOSIA (n=67/880) experienced three-month confirmed disability progression, as measured by EDSS, similar to IFN beta-1a (7.8%; n=69/889) (P=nonsignificant)).22,23
Post-hoc analysis of the DAYBREAK open label extension study (patients who completed SUNBEAM and enrolled in DAYBREAK) showed 74% of patients receiving ZEPOSIA 0.92 mg were categorized as improved or remained stable in cognitive process speed as measured by the SDMT at 60 months of treatment.26,*Score change of greater than or equal to four points was considered clinically meaningful.
ZEPOSIA has a well-established safety profile.‡ In RMS, for clinical trials, as of May 19, 2023, and in the post-marketing setting, as of April 30, 2023, ZEPOSIA has approximately 40,000 patient years of total exposure.27
Safety was comparable to AVONEX in overall incidence of adverse events in clinical trials. Overall incidence of adverse reactions for ZEPOSIA vs AVONEX at one year was 59.8% and 75.5%, respectively, and at two years was 74.7% and 83.0%, respectively. Across two head-to-head trials, the most common adverse reactions with an incidence of at least 2% in patients treated with ZEPOSIA and at least 1% greater than AVONEX, respectively, were as follows: upper respiratory infectiona, 26% (vs 23%); hepatic transaminase elevationb, 10% (vs 5%); orthostatic hypotension, 4% (vs 3%); urinary tract infection, 4% (vs 3%); back pain, 4% (vs 3%); hypertensionc, 4% (vs 2%); and upper abdominal pain, 2% (vs 1%). Data are not an adequate basis for comparison of rates between ZEPOSIA and the active control.
Severe adverse reactions: The rate of severe adverse reactions at one year for ZEPOSIA was 1.6% vs 2.2% for AVONEX and the rate at two years for ZEPOSIA was 3.5% vs 4.3% for AVONEX.
Serious adverse reactions: The rate of serious adverse reactions at one year for ZEPOSIA was 2.9% vs 2.5% for AVONEX and the rate at two years for ZEPOSIA was 6.5% vs 6.4% for AVONEX.22,23,28 Please see the full Prescribing Information for additional SUNBEAM and RADIANCE data.
Generally similar safety has been seen in the ongoing long-term extension study; nearly 10 years† of exposure.22,23,28,29
*Categorical analysis of clinically meaningful change in SDMT relative to SUNBEAM baseline. Represents participants receiving ZEPOSIA 0.92 mg who completed the SUNBEAM Phase 3 trial and enrolled in the open-label extension study.
‡ZEPOSIA has been studied in clinical trials; SUNBEAM (Phase 3) and RADIANCE (Phase 3). 882 patients receiving the 0.92 mg dose of ZEPOSIA orally once daily in SUNBEAM or RADIANCE were assessed in the safety analysis.
aUpper respiratory infection includes nasopharyngitis, upper respiratory tract infection, pharyngitis, respiratory tract infection, bronchitis, rhinitis, respiratory tract infection viral, viral upper respiratory tract infection, rhinorrhea, tracheitis and laryngitis.
bHepatic transaminase elevation includes alanine aminotransferase increased, gamma-glutamyl transferase increased, aspartate aminotransferase increased, hepatic enzyme increased, liver function test abnormal and transaminases increased.
cHypertension includes hypertension, essential hypertension and orthostatic hypertension.
†From the first patient randomized (October 18, 2012) through the DAYBREAK data cutoff (February 1, 2022), the maximum continuous exposure was 110.8 months. The mean exposure to ZEPOSIA 0.92 mg in the parent trials and DAYBREAK was 70.4 months.25
INDICATIONS
ZEPOSIA®(ozanimod)is indicated for the treatment of:
- Relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
- Moderately to severely active ulcerative colitis (UC) in adults.
ZEPOSIA 0.92 mg capsules are for oral use.
IMPORTANT SAFETY INFORMATION
Contraindications:
- Patients who in the last 6 months, experienced myocardial infarction, unstable angina, stroke, transient ischemic attack (TIA), decompensated heart failure requiring hospitalization, or Class III/IV heart failure or have a presence of Mobitz type II second-degree or third-degree atrioventricular (AV) block, sick sinus syndrome, or sino-atrial block, unless the patient has a functioning pacemaker
- Patients with severe untreated sleep apnea
- Patients taking a monoamine oxidase (MAO) inhibitor
Infections: ZEPOSIA may increase the susceptibility to infections. Life-threatening and rare fatal infections have occurred in patients receiving ZEPOSIA. Obtain a recent (i.e., within 6 months or after discontinuation of prior MS or UC therapy) complete blood count (CBC) including lymphocyte count before initiation of ZEPOSIA. Delay initiation of ZEPOSIA in patients with an active infection until the infection is resolved. Consider interruption of treatment with ZEPOSIA if a patient develops a serious infection. Continue monitoring for infections up to 3 months after discontinuing ZEPOSIA.
- Herpes zoster was reported as an adverse reaction in ZEPOSIA-treated patients. Herpes simplex encephalitis and varicella zoster meningitis have been reported with sphingosine 1-phosphate (S1P) receptor modulators. Patients without a healthcare professional-confirmed history of varicella (chickenpox), or without documentation of a full course of vaccination against varicella zoster virus (VZV), should be tested for antibodies to VZV before initiating ZEPOSIA. A full course of vaccination for antibody-negative patients with varicella vaccine is recommended prior to commencing treatment with ZEPOSIA.
- Cases of fatal cryptococcal meningitis (CM) were reported in patients treated with another S1P receptor modulator. If CM is suspected, ZEPOSIA should be suspended until cryptococcal infection has been excluded. If CM is diagnosed, appropriate treatment should be initiated.
- In the MS and UC clinical studies, patients who received ZEPOSIA were not to receive concomitant treatment with antineoplastic, non-corticosteroid immunosuppressive, or immune-modulating therapies used for treatment of MS and UC. Concomitant use of ZEPOSIA with any of these therapies would be expected to increase the risk of immunosuppression. When switching to ZEPOSIA from immunosuppressive medications, consider the duration of their effects and their mode of action to avoid unintended additive immunosuppressive effects.
- Use of live attenuated vaccines should be avoided during and for 3 months after treatment with ZEPOSIA. If live attenuated vaccine immunizations are required, administer at least 1 month prior to initiation of ZEPOSIA.
Progressive Multifocal Leukoencephalopathy (PML): PML is an opportunistic viral infection of the brain that typically occurs in patients who are immunocompromised, and that usually leads to death or severe disability.
PML has been reported in patients treated with S1P receptor modulators, including ZEPOSIA, and other MS and UC therapies and has been associated with some risk factors. If PML is suspected, withhold ZEPOSIA and perform an appropriate diagnostic evaluation.
If confirmed, treatment with ZEPOSIA should be discontinued.
Immune reconstitution inflammatory syndrome (IRIS) has been reported in MS patients treated with S1P receptor modulators who developed PML and subsequently discontinued treatment. IRIS presents as a clinical decline in the patient’s condition that may be rapid, can lead to serious neurological complications or death, and is often associated with characteristic changes on MRI. The time to onset of IRIS in patients with PML was generally within a few months after S1P receptor modulator discontinuation. Monitoring for development of IRIS and appropriate treatment of the associated inflammation should be undertaken.
Bradyarrhythmia and Atrioventricular Conduction Delays: Since initiation of ZEPOSIA may result in a transient decrease in heart rate and atrioventricular conduction delays, dose titration is recommended to help reduce cardiac effects. Initiation of ZEPOSIA without dose escalation may result in greater decreases in heart rate. If treatment with ZEPOSIA is considered, advice from a cardiologist should be sought for those individuals:
- with significant QT prolongation
- with arrhythmias requiring treatment with Class 1a or III anti-arrhythmic drugs
- with ischemic heart disease, heart failure, history of cardiac arrest or myocardial infarction, cerebrovascular disease, and uncontrolled hypertension
- with a history of Mobitz type II second-degree or higher AV block, sick sinus syndrome, or sino-atrial heart block
Liver Injury: Elevations of aminotransferases may occur in patients receiving ZEPOSIA. Obtain liver function tests, if not recently available (i.e., within 6 months), before initiation of ZEPOSIA. Patients who develop symptoms suggestive of hepatic dysfunction should have hepatic enzymes checked and ZEPOSIA should be discontinued if significant liver injury is confirmed.
Fetal Risk: There are no adequate and well-controlled studies in pregnant women. Based on animal studies, ZEPOSIA may cause fetal harm. Women of childbearing potential should use effective contraception to avoid pregnancy during treatment and for 3 months after stopping ZEPOSIA. Women who become pregnant while taking ZEPOSIA for MS may enroll in the ZEPOSIA pregnancy registry by calling 1-877-301-9314 or visiting www.zeposiapregnancyregistry.com.
Increased Blood Pressure: Increase in systolic pressure was observed after about 3 months of treatment and persisted throughout treatment. Blood pressure should be monitored during treatment and managed appropriately. Certain foods that may contain very high amounts of tyramine could cause severe hypertension in patients taking ZEPOSIA. Patients should be advised to avoid foods containing a very large amount of tyramine while taking ZEPOSIA.
Respiratory Effects: ZEPOSIA may cause a decline in pulmonary function. Spirometric evaluation of respiratory function should be performed during therapy, if clinically indicated.
Macular Edema: S1P modulators have been associated with an increased risk of macular edema. Patients with a history of uveitis or diabetes mellitus are at increased risk. Patients with a history of these conditions should have an ophthalmic evaluation of the fundus, including the macula, prior to treatment initiation and regular follow-up examinations. An ophthalmic evaluation is recommended in all patients at any time if there is a change in vision. Continued use of ZEPOSIA in patients with macular edema has not been evaluated; potential benefits and risks for the individual patient should be considered if deciding whether ZEPOSIA should be discontinued.
Posterior Reversible Encephalopathy Syndrome (PRES): Rare cases of PRES have been reported in patients receiving a S1P receptor modulator. If a ZEPOSIA-treated patient develops unexpected neurological or psychiatric symptoms or any symptom/sign suggestive of an increase in intracranial pressure, a complete physical and neurological examination should be conducted. Symptoms of PRES are usually reversible but may evolve into ischemic stroke or cerebral hemorrhage. Delay in diagnosis and treatment may lead to permanent neurological sequelae. If PRES is suspected, treatment with ZEPOSIA should be discontinued.
Unintended Additive Immunosuppressive Effects From Prior Immunosuppressive or Immune-Modulating Drugs: When switching from drugs with prolonged immune effects, the half-life and mode of action of these drugs must be considered to avoid unintended additive immunosuppressive effects while at the same time minimizing risk of disease reactivation. Initiating treatment with ZEPOSIA after treatment with alemtuzumab is not recommended.
Severe Increase in Multiple Sclerosis (MS) Disability After Stopping ZEPOSIA: In MS, severe exacerbation of disease, including disease rebound, has been rarely reported after discontinuation of a S1P receptor modulator. The possibility of severe exacerbation of disease should be considered after stopping ZEPOSIA treatment so patients should be monitored upon discontinuation. After stopping ZEPOSIA in the setting of PML, monitor for development of immune reconstitution inflammatory syndrome (PML-IRIS).
Immune System Effects After Stopping ZEPOSIA: After discontinuing ZEPOSIA, the median time for lymphocyte counts to return to the normal range was 30 days with approximately 90% of patients in the normal range within 3 months. Use of immunosuppressants within this period may lead to an additive effect on the immune system, therefore caution should be applied when initiating other drugs 4 weeks after the last dose of ZEPOSIA.
Most Common Adverse Reactions that occurred in the MS clinical trials of ZEPOSIA-treated patients (≥4%): upper respiratory infection, hepatic transaminase elevation, orthostatic hypotension, urinary tract infection, back pain, and hypertension.
In the UC clinical trials, the most common adverse reactions that occurred in ≥4% of ZEPOSIA-treated patients and greater than in patients who received placebo were upper respiratory infection, liver test increased, and headache.
Use in Specific Populations: Hepatic Impairment: Dosage adjustment in patients with mild or moderate hepatic impairment (Child-Pugh class A or B) is required, and use of ZEPOSIA in patients with severe hepatic impairment (Child-Pugh class C) is not recommended.
For additional safety information, please see the full Prescribing Information and Medication Guide.
References:
- MS International Federation. Atlas of MS Edition. (title in italics) 3rd ed. MS International Federation;2020. Accessed: August 22, 2023. https://www.msif.org/wp-content/uploads/2020/12/Atlas-3rd-Edition-Epidemiology-report-EN-updated-30-9-20.pdf
- Souza A, Kelleher A, Cooper R, Cooper RA, Iezzoni LI, Collins DM. Multiple sclerosis and mobility-related assistive technology: systematic review of literature. J Rehabil Res Dev. 2010;47(3):213-223. doi:10.1682/jrrd.2009.07.0096
- Chan A, Heck C. Mobility in multiple sclerosis: More than just a physical problem. International Journal of MS Care. 2000;2(1):51-61. doi:10.7224/1537-2073-2.1.51
- Kister I, Chamot E, Salter AR, Cutter GR, Bacon TE, Herbert J. Disability in multiple sclerosis: a reference for patients and clinicians. Neurology. 2013;80(11):1018-1024. doi:10.1212/WNL.0b013e3182872855
- Oreja-Guevara C, Ayuso Blanco T, Brieva Ruiz L, Hernández Pérez MÁ, Meca-Lallana V, Ramió-Torrentà L. Cognitive Dysfunctions and Assessments in Multiple Sclerosis. Front Neurol. 2019;10:581. Published 2019 Jun 4. doi:10.3389/fneur.2019.00581
- Batista S, Zivadinov R, Hoogs M, et al. Basal ganglia, thalamus and neocortical atrophy predicting slowed cognitive processing in multiple sclerosis. J Neurol. 2012;259(1):139-146. doi:10.1007/s00415-011-6147-1
- Julian LJ. Cognitive functioning in multiple sclerosis. Neurol Clin. 2011;29(2):507-525. doi:10.1016/j.ncl.2010.12.003
- Rudick RA, Lee JC, Nakamura K, Fisher E. Gray matter atrophy correlates with MS disability progression measured with MSFC but not EDSS. J Neurol Sci. 2009;282(1-2):106-111. doi:10.1016/j.jns.2008.11.018
- Fisher E, Lee JC, Nakamura K, Rudick RA. Gray matter atrophy in multiple sclerosis: a longitudinal study. Ann Neurol. 2008;64(3):255-265. doi:10.1002/ana.21436
- Kalb R, Beier M, Benedict RH, et al. Recommendations for cognitive screening and management in multiple sclerosis care. Mult Scler. 2018;24(13):1665-1680. doi:10.1177/1352458518803785
- Eijlers AJC, van Geest Q, Dekker I, et al. Predicting cognitive decline in multiple sclerosis: a 5-year follow-up study. Brain. 2018;141(9):2605-2618. doi:10.1093/brain/awy202
- Houtchens MK, Benedict RH, Killiany R, et al. Thalamic atrophy and cognition in multiple sclerosis. Neurology. 2007;69(12):1213-1223. doi:10.1212/01.wnl.0000276992.17011.b5
- Minagar A, Barnett MH, Benedict RH, et al. The thalamus and multiple sclerosis: modern views on pathologic, imaging, and clinical aspects. Neurology. 2013;80(2):210-219. doi:10.1212/WNL.0b013e31827b910b
- Calabrese M, Rinaldi F, Grossi P, Gallo P. Cortical pathology and cognitive impairment in multiple sclerosis. Expert Rev Neurother. 2011;11(3):425-432. doi:10.1586/ern.10.155
- Popescu BF, Pirko I, Lucchinetti CF. Pathology of multiple sclerosis: where do we stand?. Continuum. 2013;19(4 Multiple Sclerosis):901-921. doi:10.1212/01.CON.0000433291.23091.65
- Crespy L, Zaaraoui W, Lemaire M, et al. Prevalence of grey matter pathology in early multiple sclerosis assessed by magnetization transfer ratio imaging. PLoS One. 2011;6(9):e24969. doi:10.1371/journal.pone.0024969
- Andorra M, Nakamura K, Lampert EJ, et al. Assessing Biological and Methodological Aspects of Brain Volume Loss in Multiple Sclerosis. JAMA Neurol. 2018;75(10):1246-1255. doi:10.1001/jamaneurol.2018.1596
- He A, Merkel B, Brown JWL, et al. Timing of high-efficacy therapy for multiple sclerosis: a retrospective observational cohort study. Lancet Neurol. 2020;19(4):307-316. doi:10.1016/S1474-4422(20)30067-3
- Sotirchos ES, Gonzalez-Caldito N, Dewey BE, et al. Effect of disease-modifying therapies on subcortical gray matter atrophy in multiple sclerosis. Mult Scler. 2020;26(3):312-321. doi:10.1177/1352458519826364
- Wattjes MP, Ciccarelli O, Reich DS, et al. 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis. Lancet Neurol. 2021;20(8):653-670. doi:10.1016/S1474-4422(21)00095-8
- Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33(11):1444-1452. doi:10.1212/wnl.33.11.1444
- Cohen JA, Comi G, Selmaj KW, et al. Safety and efficacy of ozanimod versus interferon beta-1a in relapsing multiple sclerosis (RADIANCE): a multicentre, randomised, 24-month, phase 3 trial. Lancet Neurol. 2019;18(11):1021-1033. doi:10.1016/S1474-4422(19)30238-8
- ZEPOSIA. Prescribing Information. Bristol-Myers Squibb Company; 2023.
- Data on File. Bristol Myers Squibb. 2023.
- Selmaj KW, Steinman L, Comi G, et al. Long-Term Safety and Efficacy of Ozanimod in Relapsing Multiple Sclerosis: Interim Analysis of the DAYBREAK Open-Label Extension Study. Poster presented at: 38th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS); October 26-28, 2022; Amsterdam, Netherlands.
- DeLuca J, Cohen JA, Cree BAC, et al. Effects of Ozanimod on Cognitive Processing Speed: Findings from the Phase 3 SUNBEAM and DAYBREAK Extension Trials. Poster presented at: Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) 2023 Forum; February 23-25, 2023; San Diego, CA, USA.
- Data on File. Bristol Myers Squibb. 2023.
- Comi G, Kappos L, Selmaj KW, et al. Safety and efficacy of ozanimod versus interferon beta-1a in relapsing multiple sclerosis (SUNBEAM): a multicentre, randomised, minimum 12-month, phase 3 trial. Lancet Neurol. 2019;18(11):1009-1020. doi:10.1016/S1474-4422(19)30239-X
- Selmaj KW, Steinman L, Comi G, et al. Long-Term Safety and Efficacy of Ozanimod in Relapsing Multiple Sclerosis: Interim Analysis of the DAYBREAK Open-Label Extension Study. Poster presented at: 37th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS); October 13-15, 2021; Virtual
Newsletter
Keep your finger on the pulse of neurology—subscribe to NeurologyLive for expert interviews, new data, and breakthrough treatment updates.