Neuromuscular
Latest News
Latest Videos

CME Content
More News

Three of 4 patients withdrew from their previous enzyme replacement therapy following treatment with AT845 gene therapy.

A group of experts in the care of patients with rare diseases—Emma Ciafaloni, MD, FAAN; Martina Bebin, MD, MDA; Timothy Miller, MD, PhD; George Small, MD; Bruce Cree, MD, PhD, MAS, FAAN—shared their perspectives on hot topics of treatment and management.

In recognition of Rare Disease Day, the NeurologyLive® team offered an extensive update on the state of care and treatment for a few rare neurological diseases, including Pompe disease, Rett syndrome, among others.

In recognition of Rare Disease Day, the NeurologyLive® team offered an extensive update on the state of care and treatment for a few rare neurological diseases, including Pompe disease, Rett syndrome, among others.

In a dose-ranging study, the gene therapy achieved 78% Gb3 substrate clearance at 6 months and 77% reduction in urine podocyte loss in 1 of the first kidney biopsies.

Here's some of what is coming soon to NeurologyLive® this week.

Take 5 minutes to catch up on NeurologyLive®'s highlights from the week ending February 24, 2023.
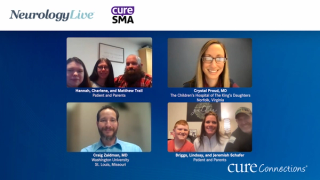
Drs Juliana Gurgel Giannetti, Rodrigo de Holanda Mendonça and Marcelo Kerstenetzky examine barriers to participation in clinical trials in Brazil for patients with spinal muscular atrophy. Drs Juliana Gurgel Giannetti, Rodrigo de Holanda Mendonça e Marcelo Kerstenetzky analisam barreiras para a participação de pacientes com atrofia muscular espinhal em testes clínicos no Brasil.

The expert panel discusses challenges in clinical monitoring and access to care for patients with spinal muscular atrophy in Brazil. O painel de especialistas discute desafios no monitoramento clínico e no acesso a atendimento de saúde para pacientes com atrofia muscular espinhal.

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.

Stanley Appel, MD, shared his insight into the MDA session track on amyotrophic lateral sclerosis, the latest science advances to benefit patients, and where the field is headed with research and treatment.

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.

Here's some of what is coming soon to NeurologyLive® this week.
The lead of the Myasthenia Gravis Foundation of America Global Myasthenia Gravis Patient Registry spoke on 2 additional attributes of the patient registry. [WATCH TIME: 3 minutes]

Take 5 minutes to catch up on NeurologyLive®'s highlights from the week ending February 17, 2023.

The assistant professor of neurology at the Yale School of Medicine spoke about the strength in the patient-clinician relationship in ensuring quality care for myasthenia gravis. [WATCH TIME: 4 minutes]

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.

In honor of Duchenne Muscular Dystrophy Awareness Week, held February 13, 2023, to February 19, 2023, get caught up on some of the latest news in Duchenne muscular dystrophy as the NeurologyLive® team shares some of our data updates.

Drs Juliana Gurgel Giannetti, Rodrigo de Holanda Mendonça and Adriana Banzzatto Ortega consider best practices for management based on the type of spinal muscular atrophy, age, and the therapy received. Drs Juliana Gurgel Giannetti, Rodrigo de Holanda Mendonça e Adriana Banzzatto Ortega consideram melhores práticas de conduta baseados no tipo de atrofia muscular espinhal, idade e terapia recebida.

The expert panel discusses management of family expectations regarding post-therapy outcomes and strict patient monitoring. O painel de especialistas discute como lidar com expectativas das famílias em relação a resultados terapêuticos e em períodos de monitoramento rigoroso.

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.

A meta-analysis displayed a small magnitude of pooled effect estimates for the studied skip-amenable mutation classes in Duchenne muscular dystrophy at 1 year follow up.

Dual-task effects were greater for patients with functional gait disorders than controls in gait speed and stride length, but not in stride time variability.

To honor Black History Month, NeurologyLive® spoke with influential Black clinicians on the leaders they look up to, the ongoing fight to overcome racial disparities, and ways to encourage diversity in health care.